- Home
- Publications
- PAGES Magazine
- A Whole Ocean Thermometer From Atmospheric Noble Gas Ratios
A whole ocean thermometer from atmospheric noble gas ratios
Sarah Shackleton
Past Global Changes Magazine
31(2)
96-97
2023
Marine-based reconstructions of ocean temperature have provided fundamental insight into past climate. The novel ice-core based proxy for mean ocean temperature from atmospheric noble gas ratios has demonstrated promise in furthering these insights.
Modern ocean warming
The oceans represent the largest source of thermal inertia in the climate system and play a key role in modulating the pacing of climate change. Measuring how much heat the oceans have taken up since the onset of the industrial era is important for understanding how the planet has responded to changes in our atmospheric composition driven by human activity. However, measuring total ocean warming is not an easy task, as heat uptake is spatially heterogeneous. Until recently, measurements of much of the ocean’s temperature have been sparse, and few measurements exist of ocean temperature below 2000 meters, where almost half of the ocean’s volume resides.
Probing ocean-temperature change before the instrumental era is an even greater challenge. Most of our information about past ocean temperature comes from marine-sediment records. However, as in the case of the instrumental era, ocean-temperature change at one location does not necessarily give you information about the global trend. In addition, most of our marine-sediment proxies for temperature provide information about sea-surface temperature, which represents a tiny fraction of the total ocean volume.
Changes in ocean temperature can also change its composition, including the quantity of gases dissolved in seawater. As the ocean warms, it can hold less gas, which leads to a net degassing of seawater as it surfaces and warms. This has important consequences for our atmosphere and climate; as seawater warms it can take up less CO2, leading to an increase in atmospheric CO2 and further warming.
Inert atmospheric gases trace ocean heat
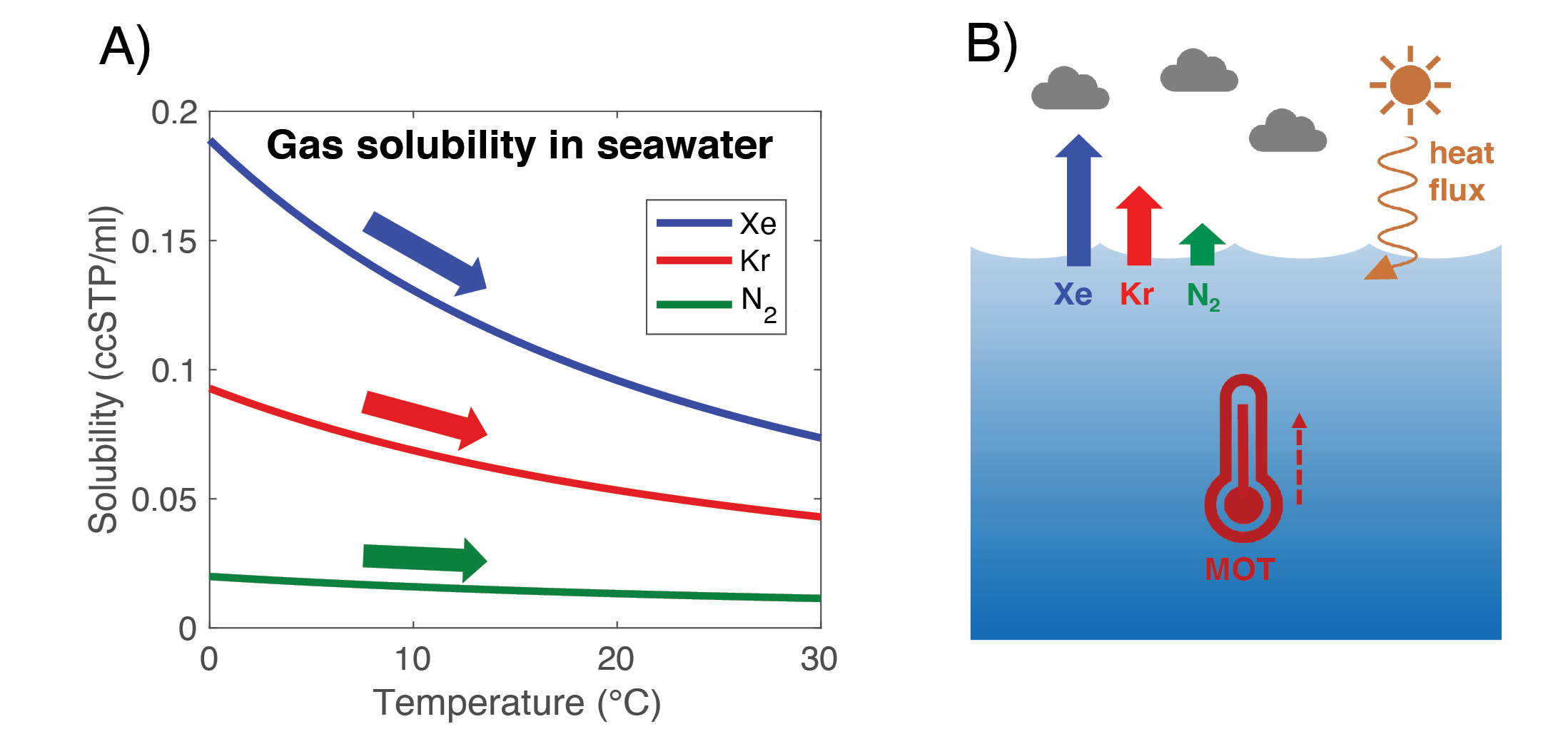
This temperature dependence of gas solubility in seawater also has consequences for the inert (or non-reactive) noble gases, including krypton (Kr) and xenon (Xe). The larger the noble gas, the higher its solubility in seawater, and the stronger the temperature dependence of that solubility (Fig. 1).
 |
|
Figure 1: (A) Temperature dependence of inert gas solubilities in seawater (35 practical salinity units) and (B) schematic of the noble gas mean ocean temperature (MOT) proxy. |
Between the ocean and atmosphere, about 5% of xenon is dissolved in the ocean and 95% resides in the atmosphere. At the average ocean temperature (3.5°C), the solubility of xenon changes by about 4% per °C of warming. Therefore, if the whole ocean warmed by 1°C, the concentration of xenon in the atmosphere would increase by roughly 0.2%. This may sound like a tiny change, especially given that xenon has an atmospheric concentration of only 87 parts per billion. However, these small changes may be measured.
In ice cores, we can measure the ratios of xenon and krypton in air bubbles with respect to one another (Xe/Kr) or relative to N2 (Xe/N2 and Kr/N2) to reconstruct global mean ocean temperature (Headly and Severinghaus 2007). While N2 is not entirely inert and may be converted to bio-available forms via nitrogen fixation, this bio-available nitrogen represents a miniscule portion (<0.01%) of the total nitrogen in the ocean and atmosphere; even a dramatic change to the nitrogen cycle would lead to a negligible change in the total N2 inventory. We may therefore treat N2 as an inert tracer, as we do Xe and Kr.
Why is this a whole ocean thermometer?
As implied above, seawater will only degas or ingas when it is at the surface and may equilibrate with the atmosphere, so it is not necessarily intuitive why the atmospheric noble gas ratios reflect mean ocean temperature change rather than that of the surface. To gain insight, first we must understand that all of the ocean’s temperature (including water at depth) is set at the sea surface. Geothermal heating may warm the ocean from below, but this term is miniscule in comparison to the ocean–atmosphere heat flux. When a parcel of water is cooled at the surface it will take up more noble gases before it sinks into the deep. Once at depth, it conserves these properties (heat and inert gases), which are only reset once the parcel resurfaces. This means that at any given time, the total sum of dissolved xenon, krypton, and nitrogen in all these parcels of water (which controls atmospheric Kr/N2, Xe/N2, and Xe/Kr) should reflect total heat content. Because the atmosphere is mixed on annual timescales, the atmospheric noble gas ratios track ocean temperature change with no lag.
What can we learn?
Only a small number of studies have been published using this noble-gas-based technique in ice cores. However, they have shown the great potential of this proxy in probing the dynamic relationships between ocean-heat content and other components of our climate system.
-
Ocean circulation and ocean-heat content
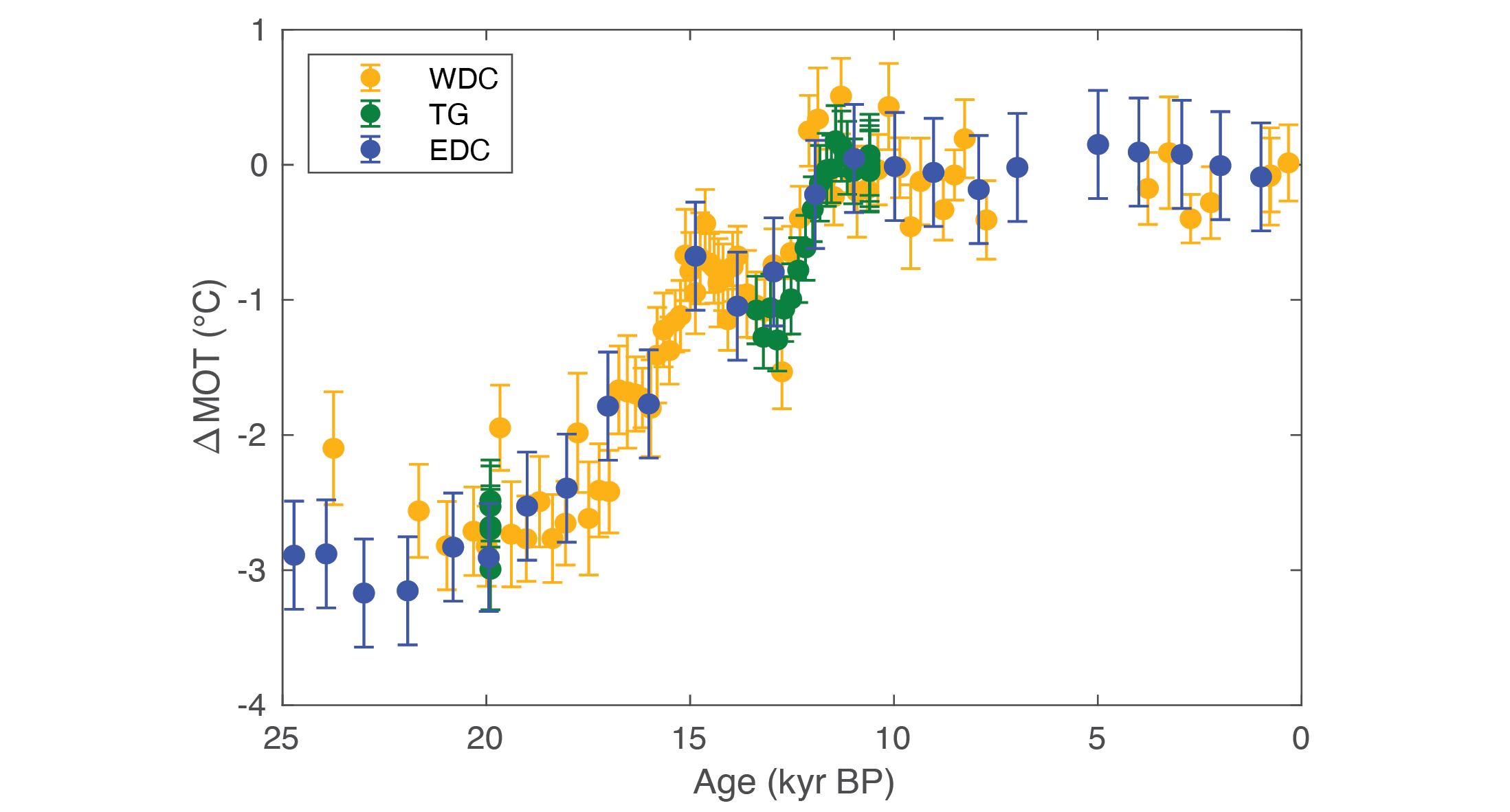
The first timeseries of mean ocean temperature came from the West Antarctic Ice Sheet (WAIS) Divide ice core and covered the most recent transition between glacial and interglacial climate (i.e. the last 25,000 years; Bereiter et al. 2018). This transition involved mean ocean warming in two pronounced steps, both of which occurred during millennial-scale disruptions in ocean overturning. This was later confirmed by two other reconstructions of mean ocean temperature over this interval (Baggenstos et al. 2019; Shackleton et al. 2019; Fig. 2) and suggests an important link between ocean circulation and heat content.
-
CO2 and ocean temperature
The relationship between CO2 and ocean temperature is bi-directional; CO2 warms our planet (thus warming the oceans), and due to reduced solubility, warmer oceans can hold less CO2, thereby increasing atmospheric CO2. The noble gas proxy for mean ocean temperature is well suited to probe this relationship, because the noble gases and CO2 are measured on the same archive, which makes it possible to reconstruct changes in ocean temperature and CO2 without uncertainty in the relative timing of these changes.
The links between ocean temperature and atmospheric CO2 have been probed in several recent studies. Shackleton et al. (2021) examined the role of ocean cooling in lowering atmospheric CO2 during an interval of abrupt CO2 drawdown in the last glacial cycle. Haeberli et al. (2021) evaluated the relationship between atmospheric CO2 and climate on orbital timescales by assessing mean ocean temperature and CO2 records during the interglacials and glacial maxima of the last 700,000 years.
-
Ocean temperature, sea level, and ice volume
Ocean warming contributes directly to sea-level rise through the thermal expansion of seawater. Mean ocean–temperature reconstructions may be used to quantify this direct role of ocean warming in contributing elevated sea levels during part warm intervals, such as the last interglacial (Shackleton et al. 2020).
Ice-core noble gases may provide additional constraints on past sea-level and ice volume through insights into records of the oxygen isotopic composition (δ18O) of benthic foraminifera, which is set by ocean temperature and seawater δ18O. As mean seawater δ18O is controlled by the growth and decay of 18O-depleted ice sheets, global composites of benthic-foraminiferal δ18O record changes in mean ocean temperature and global ice volume. Insight into the relative influences of ocean temperature and ice volume on δ18O may therefore be gained by comparing contemporaneous records of global δ18O and mean ocean temperature.
Conclusions
While mean ocean–temperature reconstructions in ice cores have shown promising results and offered new paleoclimatic insight, there is plenty to learn about noble-gas-based tools. Questions remain about the potential complexities of the proxy and ways in which the noble gases measured in ice cores may become decoupled from ocean-heat content. In the future, as much attention should be put into understanding the potential pitfalls of the proxy as in producing new records.
affiliation
Department of Geosciences, Princeton University, USA
contact
Sarah Shackleton: ss77 princeton.edu
princeton.edu
references
Baggenstos D et al. (2019) Proc Natl Acad Sci U S A 116: 14881-14886
Bereiter B et al. (2018) Nature 553: 39-44
Haeberli M et al. (2021) Clim Past 17: 843-867
Headly MA, Severinghaus JP (2007) J Geophys Res 112: D19105
Shackleton S et al. (2019) Geophys Res Lett 46: 5928-5936