- Home
- Publications
- PAGES Magazine
- Radiocarbon Dating of Alpine Ice Cores
Radiocarbon dating of alpine ice cores
Ling Fang, T.M. Jenk and M. Schwikowski
Past Global Changes Magazine
31(2)
106-107
2023
An accurate chronology of alpine ice cores is essential to interpret the climate-signal and atmospheric-pollution history archived in glaciers. The radiocarbon in water-insoluble organic carbon (WIOC) has emerged as a valuable tool for dating alpine ice cores.
The challenge of alpine ice-core dating
The most common ice-core dating approach is annual layer counting, which relies on seasonal variations in chemical and physical signals, such as ammonium, stable isotope ratios (δ18O, δ2H) and solid electrical conductivity. However, pronounced thinning beyond a certain depth limits this approach in its application for ice cores.
To establish a complete age–depth scale down to the bedrock, simplified ice-flow models can be employed (e.g. Dansgaard and Johnsen 1969), but they are unable to resolve small-scale variations in ice flow (i.e. thinning/strain), particularly closer to the bedrock, where the often complex glacier geometries of high-mountain glaciers become increasingly important. Also, they rely on fundamental assumptions, such as constant accumulation, that likely do not reflect the actual conditions over time. Even complex 3D models cannot convincingly simulate the age of the deepest sections, if no additional age constraints are available (e.g. Licciulli et al. 2020).
Absolute time horizons can pin down the age of the ice. A valuable marker for alpine ice cores is the signal from atmospheric nuclear-weapon testing, showing up as a clear peak in 1963 CE in multiple proxies, such as tritium or cesium-137. Known volcanic eruptions, such as Katmai in 1912 CE and Tambora in 1815 CE, indicated by peaks in sulfate and conductivity, are also commonly used as time markers (Herren et al. 2013). Moreover, Saharan dust events during the 20th century (e.g. 1977, 1947 and 1901 CE) are well documented and can easily be identified in the European Alps (often visually, coinciding with peaks in calcium concentration in the ice cores). However, these events were only documented for the last two centuries, and these horizons cannot be established in deeper sections where no conventional dating techniques are applicable.
The development of radiocarbon analysis of WIOC
Radioactive nuclides entrapped in the ice offer an opportunity to obtain absolute dates. The environmental radionuclide 210Pb, with a short half-life of 22.3 years, enables the dating of ice over roughly one to two centuries (Gäggeler et al. 2020). The noble gas 39Ar and 32Si with a half-life of 268 ± 8 years and of 144 ± 11 years, respectively, have been demonstrated as ideal dating isotopes for ice samples from the last thousand years (Morgenstern et al. 2010; Ritterbusch et al. 2022).
The recent developments in atom-trap trace analysis (ATTA) have allowed scientists to substantially reduce the required amount of ice to 1–3 kg for 39Ar dating (Ritterbusch et al. 2022). The long-lived 81Kr, with a half-life of 229,000 years, can date ice up to 1.5 million years old (Tian et al. 2019). However, due to the low abundance of 81Kr, ~10 kg of Antarctic ice or 20–40 kg of ice from the Tibetan Plateau is recommended for sampling (Tian et al. 2019). Given the half-life of 5370 years, radiocarbon (14C) is considered the most suitable radionuclide for dating ice samples up to around 20,000 years old, covering most of the time range typically accessible by alpine ice cores (Uglietti et al. 2016).
Previously, 14C dating of ice was only possible where sufficient organic matter such as plant, wood or insect fragments was found (Thompson et al. 1998). However, the occurrence of such findings is rare in glacier ice, and even when they are present, do not allow for continuous dating. To overcome this challenge, Jenk et al. (2009) introduced the use of water insoluble organic carbon (WIOC) for 14C dating of alpine ice cores and Uglietti et al. (2016) later validated the method.
Carbonaceous particles are a major component of the atmospheric aerosol and deposit onto the glacier by precipitation. They are composed of two main bulk fractions: organic carbon (OC) and elemental carbon (EC). OC can be split into WIOC and dissolved organic carbon (DOC; see below) by solubility. WIOC and EC are separated based on their specific thermal properties (combustion temperatures). The micro-carbon 14C dating method relies on the finding that WIOC originated solely from biosphere emissions prior to the use of fossil fuels (~1850 CE; Jenk et al. 2006). Once emitted, the 14C decays according to its half-life time, starting the radiometric clock. A detailed method description for WIOC 14C-dating can be found in Uglietti et al. (2016).
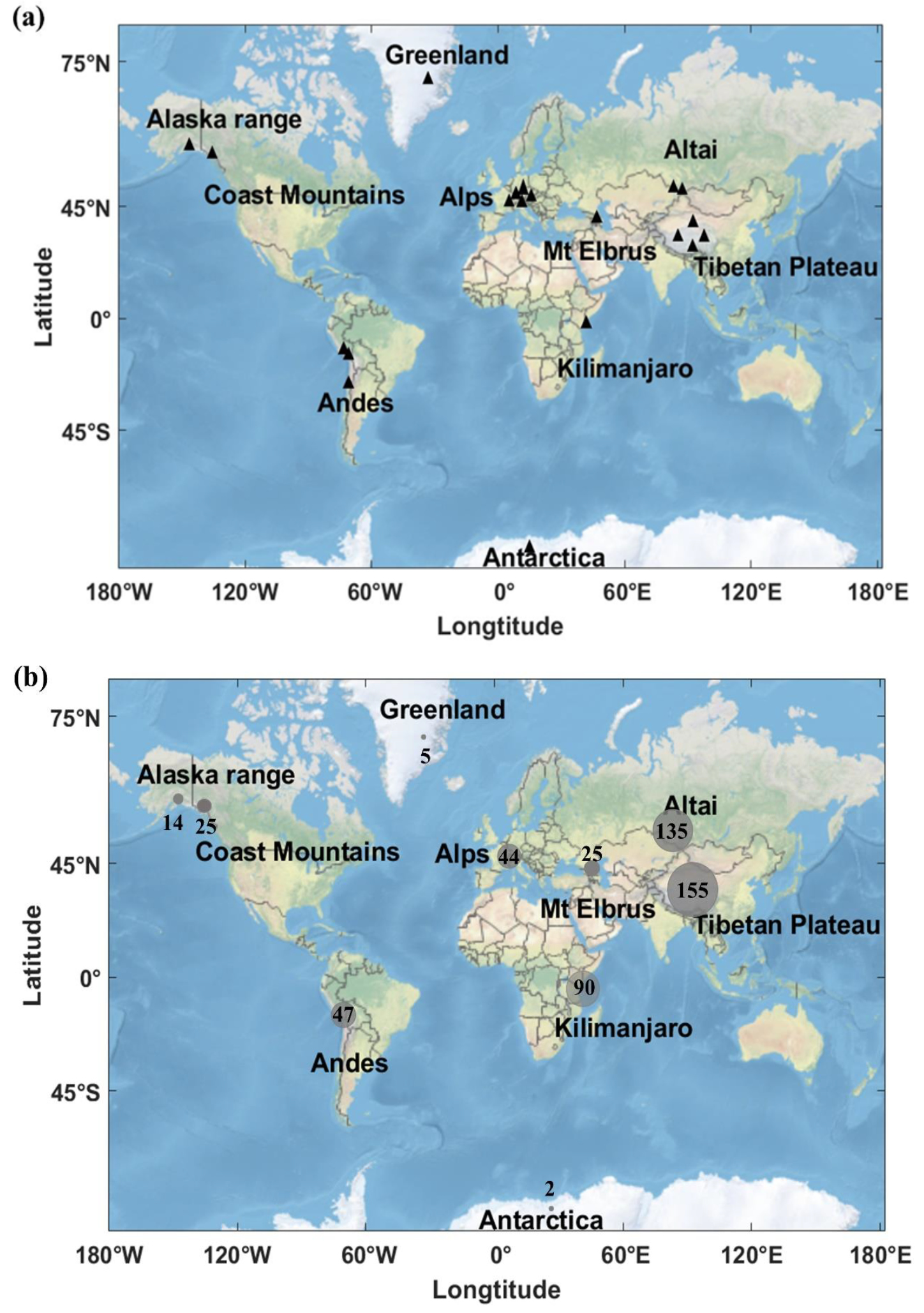
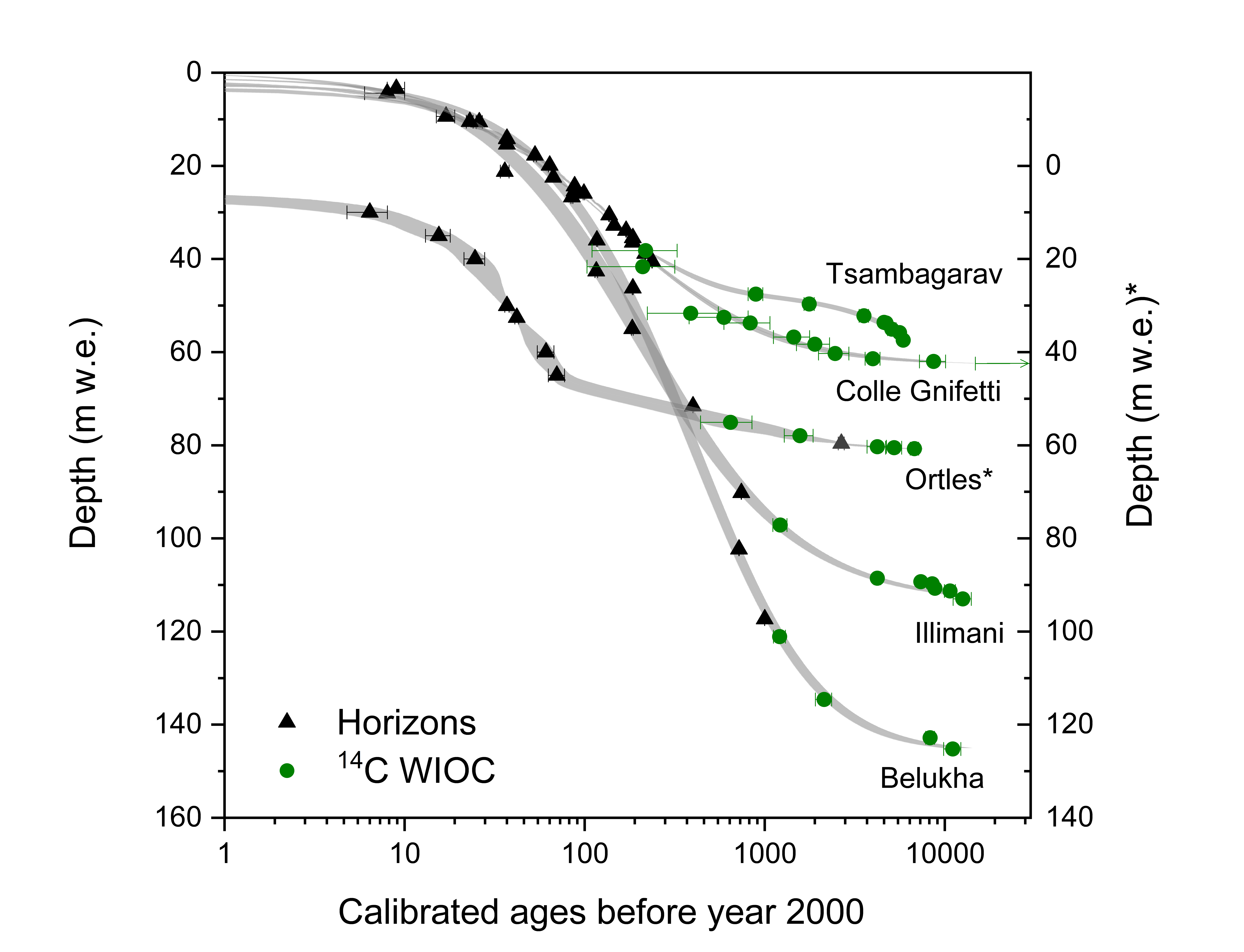
Various ice samples have been dated using micro-14C WIOC since the introduction of this technique (Figs. 1–2). WIOC concentrations ranged from 2–15 μg/kg in samples from the Polar Regions to 155 μg/kg in ice from the Tibetan Plateau (Fang et al. 2021; Uglietti et al. 2016), while Alpine ice samples used for dating contained 44 ± 14 μg/kg on average (Fang et al. 2021; Uglietti et al. 2016; Fig. 1). To date, the oldest sample was determined with an age of ~22 kyr BP at the bottom of an ice core retrieved from Belukha (Russian Altai; Fig. 2). On Colle Gnifetti (CG03 core) in the European Alps, the oldest ice retrieved from the Alps was >15 kyr BP, and on Illimani in the Andes ~12.6 kyr BP (Fang et al. 2021; Jenk et al. 2009; Sigl et al. 2009; Uglietti et al. 2016; Fig. 2).
The majority of dated alpine ice cores, however, was younger than 10 kyr BP (e.g. Uglietti et al. 2016). The dating precision strongly depends on the 14C content of the sample, defined by the carbon mass and the age of the sample, i.e. its 14C/12C ratio. The uncertainty decreases sharply with increased carbon mass due to the blank correction. Therefore, a total carbon amount of 10 μg, typically equivalent to around 300–500 g of ice, is recommended for reliable dating.
The most recent developments and outlook
In glacier ice, the higher concentration of DOC compared to WIOC (by a factor ranging from 2 to 5; Fang et al. 2021) provides a motivation to investigate the possibility of using DOC for 14C dating. The required mass of ice could potentially be further reduced, if WIOC and DOC were to be extracted from the same piece of ice. Two studies found that the DOC fraction can be biased in its 14C/12C ratio due to in situ 14C production by cosmogenic radiation (Fang et al. 2021; May 2009). This limits the theoretically achievable gain in dating precision given by the higher carbon mass.
Nevertheless, Fang et al. (2021) showed the great potential of the DOC fraction to date ice from sites where in situ 14C production is relatively low. This is the case for sites at altitudes of 4000–5000 masl and below (low radiation), and/or characterized by snow accumulation rates greater than 0.5–1 m water equivalent (less exposure) such as in the European Alps. Method details can be found in Fang et al. (2019), also describing the most recent setup allowing simultaneous extraction of DOC and WIOC samples for 14C dating of ice, which was built at the Laboratory for Environmental Chemistry (PSI, Switzerland).
In conclusion, due to lower demands of ice mass and the time coverage of the lowest and oldest ice-core sections, 14C analysis has become a crucial and widely applied tool for the dating of ice cores from mountain glaciers. Although the possibility of using the DOC fraction for ice-core dating has been demonstrated, further studies are needed to explore the full dating potential in terms of ice requirement and analytical precision, and accuracy for using both OC fractions, extracted from the same sample.
affiliationS
1Laboratory for Environmental Chemistry, Paul Scherrer Institute, Villigen, Switzerland
2Oeschger Centre for Climate Change Research, University of Bern, Switzerland
3College of Urban and Environmental Science, Northwest University, Xi’an, China
4Department of Chemistry and Biochemistry, University of Bern, Switzerland
contact
Ling Fang: lingfang nwu.edu.cn
nwu.edu.cn
references
Dansgaard W, Johnsen SJ (1969) J Glaciol 8: 215-223
Fang L et al. (2019) Radiocarbon 61: 681-694
Fang L et al. (2021) The Cryosphere 15: 1537-1550
Gäggeler HW et al. (2020) J Glaciol 66: 447-456
Herren P-A et al. (2013) Quat Sci Rev 69: 59-68
Jenk TM et al. (2006) Atmos Chem Phys 6: 5381-5390
Jenk TM et al. (2009) J Geophys Res Atmos 114, D14305
Licciulli C et al. (2020) J Glaciol 66: 35-48
May BL (2009) PhD Thesis. University of Heidelberg, 160 pp
Morgenstern U et al. (2010) Nucl Instrum Methods Phys Res Sect B Beam Interact Mater At 268: 739-743
Ritterbusch F et al. (2022) Proc Natl Acad Sci 119: e2200835119
Sigl M et al. (2009) J Glaciol 55: 985-996
Thompson LG et al. (1998) Science 282: 1858-1864